Introduction
An immunoassay that uses radiolabeled molecules to form immune complexes step by step is referred to as a radioimmunoassay (RIA). The technique is used to measure antigen concentrations (for example, hormone levels in blood) in vitro by using antibodies. At the Veterans Administration Hospital in New York, Solomon Berson and Rosalyn Yalow first described RIA for measuring endogenous plasma insulin in 1960.
Principle
The Ag-Ab complex is created by the specific binding of antigens and antibodies. Radioisotopes can be used to mark or conjugate the antigen. The sample’s unlabeled antigens compete with radiolabeled antigens for binding sites on particular antibodies. The labelled antigens that are already connected to the antibodies are replaced with the unlabeled antigens. The amount of free radiolabeled antigens in the solution increases when the unlabeled antigens bind with antibodies. Since bound unlabeled antigens are precisely proportional to the concentration of free labelled antigens, this is the case.
Three principles are combined in its implementation:
1. An immunological response, i.e. antigen and antibody binding.
2. A reaction of competitive binding or displacement. (It provides specificity).
3. Radio emission measurement. (It demonstrates sensitivity).
Requirements
Radiolabeled antigens (or hot antigens): antigens radiolabeled typically by beta-ray emitting isotopes like tritium and gamma-ray emitting isotopes like I-125.
Specific antibodies: Needed in less amount than antigens.
Unlabeled antigens (or cold antigens): include sample antigens.
Microtitre plates: 96-well microtitre plates.
Washing Buffer solutions: like 1 % Trifluoroacetic acid.
Procedure
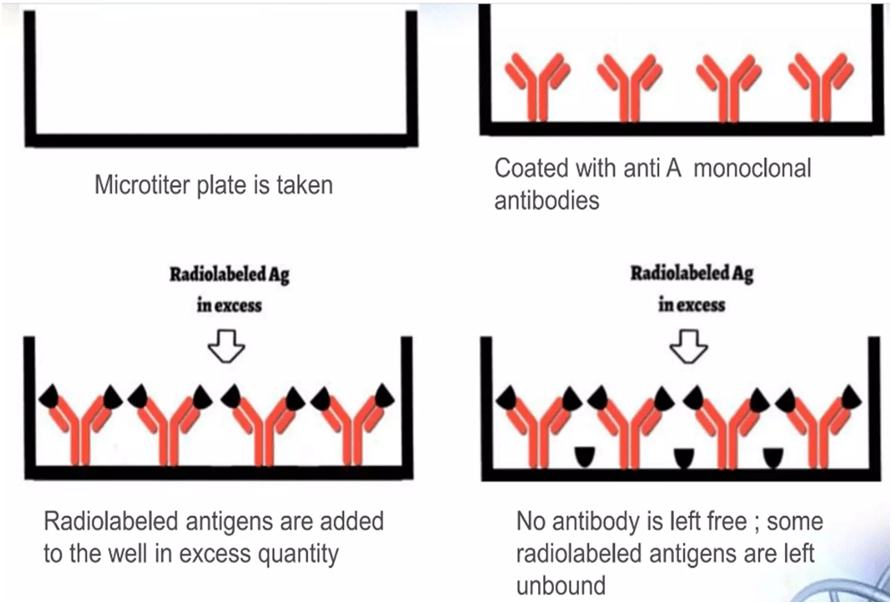
- Particular antibodies fixed in microtitre wells at defined concentrations.
- Next, a certain quantity of hot antigens is added to the well.
- Thoroughly wash to get rid of any unbound antigens.
- The well’s radioactivity will be at its highest at this point.
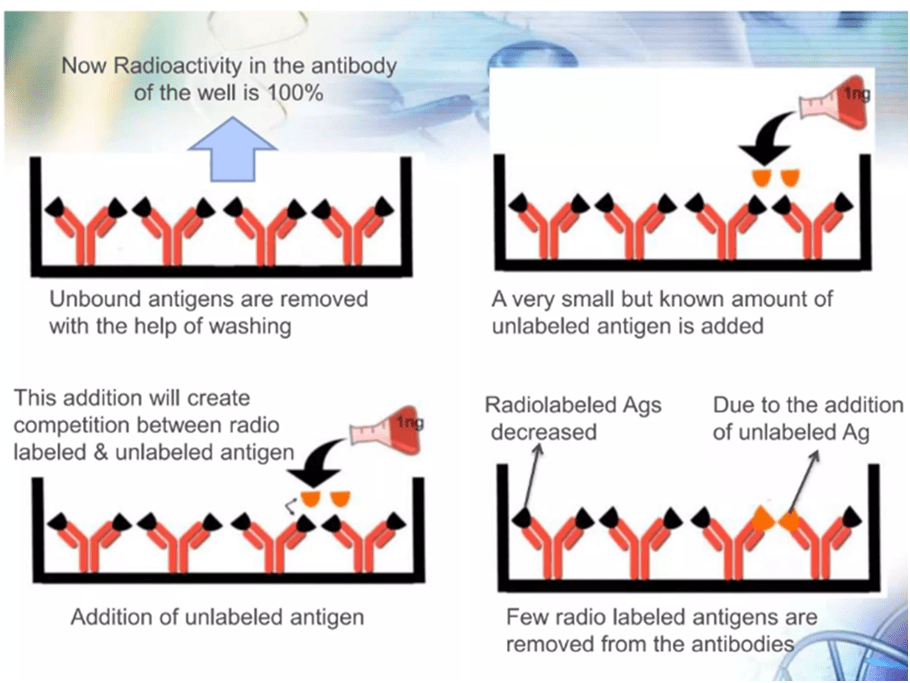
- The well is then filled with unlabeled antigens.
- Unlabeled antigens will bind to antibodies and there will be free labeled antigens in well.
- Carefully cleaned one more to get rid of free labelled antigens.
- A gamma-counter is then used to measure the radioactivity of the wells.
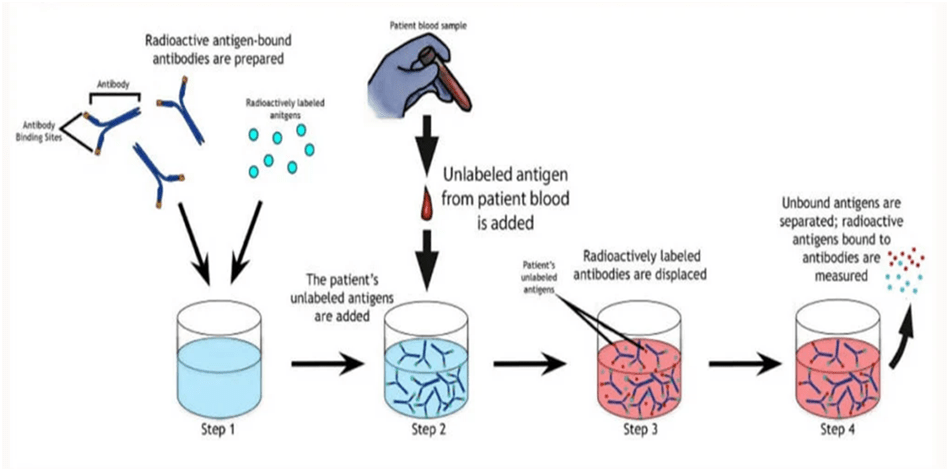
Result Interpretation
1. The radioactivity of labelled antigens will be at its peak when they first bind to antibodies.
2. If the sample includes certain antigens of interest, they will bind to antibodies and release labelled antigens, which will reduce the radioactivity of the solution.
3. As a result, the presence of the target antigen in the sample can be determined by observing a decrease in radioactivity. Additionally, it can be considered a negative test if the radioactivity stays the same.
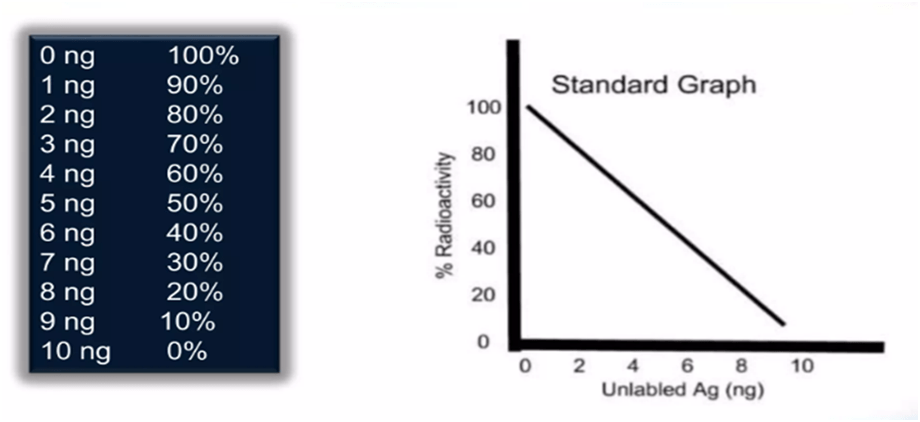
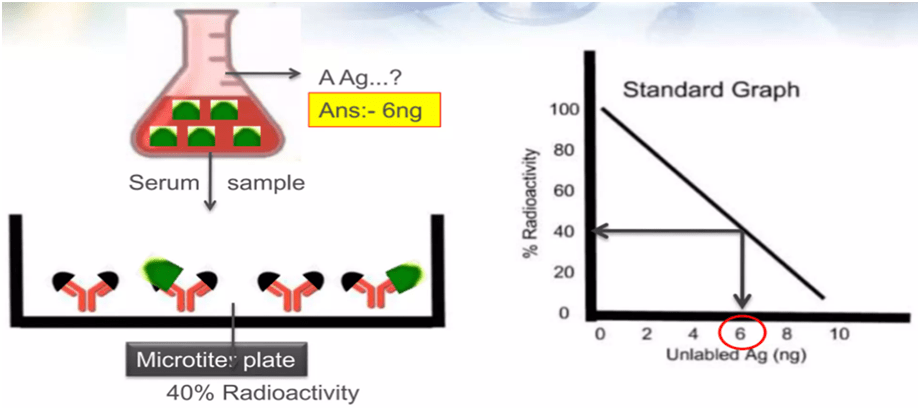
4. The radioactivity decreases as the concentration of unlabeled antigens increases. A standard curve is produced by graphing radioactivity (in percentage) vs the quantity of unlabeled antigens.
5. The method for the sample to be analysed is carried out in parallel, and the radioactivity measurement is calibrated using a reference curve to calculate the antigen concentration.
Applications
- To detect peptide hormones.
- Identification of various viral antigens.
- To detect variety of hormones and drugs.
- Hepatitis B surface antigens detection.
- To confirm presence of mycotoxins.
- Early-stagedetection of cancer.
Advantages
- High sensitivity.
- High specificity.
- Capable of detecting antibodies or antigens in small (nanogram) quantities.
Limitations
- Working with radioactive materials is a risky task.
- Disposal of radioactive materials is a problematic task.
- Reagents and equipment are costly.
- The shelf life of radiolabeled compounds is very short.