By ROBERT VAN WYHE and TIAGO TEDESCHI DOS SANTOS*
In terms of nutritional understanding, dietary fiber is recognized as the least-well-known major feed component (Angel and Sorbara, 2014).
With the decline in the use of antibiotics as growth promoters, there is more attention on the other effects of fiber, such as how it influences the as- similation of other dietary components (Aman and Graham, 1990), volatile fatty acid (VFA) production and microflora modulation. The importance of fiber in animal nutrition is attributed not only to its nutritional value but also the effects fiber quantity, composition and charac- teristics have on the absorption of other nutrients and on gut function.
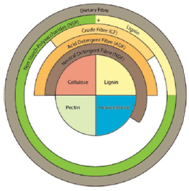
Part of the difficulty in determining the impact of fiber comes from the fact that fiber is a broad term that includes a number of different structures that are not necessarily related to each other. Biologically, dietary fiber can be defined as plant material not digested by animal digestive enzymes. Chemically, the defi- nition most widely accepted is the sum of non-starch polysaccharides (NSPs) and lignin (EU 2008/100/EG).
The fibrous component of animal di- ets has historically been analyzed and reported as crude fiber, neutral deter- gent fiber (NDF) and acid detergent fiber (ADF), where the ingredient is subjected to extraction with various neutral, acid and/or alkaline solutions (Figure 1). These methods provide a measure of the insoluble fibers present in ADF and NDF but do not give a measurement of the NSPs.
Recent advances in enzymatic and chemical methods provide information on the monomeric composition of the NSPs divided into both soluble and in- soluble fractions (Knudsen, 2001). These methods are helpful for understanding the characteristics of these fibrous com- ponents as, for example, it is recognized that a more soluble NSP component in cereals may result in higher digesta vis- cosity and, thus, reduced nutrient di- gestibility in the animal.
Determination of monomer fiber car- bohydrates should be considered with caution because the content and composition may not adequately predict their physiological effects within the animal. This happens because cell wall compo- nents are often linked, so it’s the macro- molecular complex — not the individual components — that is responsible for physiological responses (Aman and Had- den, 1990).
This is even more important when comparing the composition of different ingredients, as the fibrous structure will vary depending on origin. For example, xylose moieties in cereals such as corn and wheat will be mainly associated with arabinose, forming arabinoxylans, while in leguminous ingredients such as soy or canola, they will be primarily associated with glucans, forming xyloglucans.
The complexity of fiber determination and classification helps explain the vari- ability seen in the literature where the impact of fiber on animal nutrition has been assessed. When different compo- nents with different characteristics and different structures are classified simi- larly as “fiber,” very different effects can be expected.
NSPs are considered the biggest com- ponent in feed formulation with anti- nutritional characteristics, as they are resistant to digestive enzymes, can form viscous solutions and affect the move- ment and the digestibility of nutrients (Yegani and Korver, 2008). For example, wheat bran has been shown to reduce the dry matter digestibility and energy of broilers (Kras et al., 2013) since this type of insoluble, lignified fiber is poorly digested and acts as a dilutor of other nutrients.
However, when comparing relatively soluble (beet pulp) and insoluble (barley hull) fibrous sources, Jimenez-Moreno et al. (2009) observed an improvement in dry matter and nitrogen retention and an increase in gizzard size in animals fed the insoluble fiber source, whereas increased intestine size was observed in animals fed beet pulp. The authors suggested that the higher water-holding capacity of the soluble fiber in the beet pulp increased water retention in the digesta, generating higher pressure in the intestine and stimulating intestinal growth.
Interestingly, Sacraine et al. (2012) found that the increase in gizzard size was lost when feeding an insoluble fiber source that had been finely ground.
Dietary fiber and fiber fermentation affect the microbiome composition in the intestinal tract of animals (de Vries, 2014). Fiber is a substrate for bacte- rial growth in the hindgut (Kumar et al., 2012), with fermentation increasing VFA production. Bacterial diversity in the intestinal tract decreases with age and can be influenced by a variety of factors, such as vaccinations or dietary interven- tions (Ballou et al., 2016).
Recent research conducted by AB Vista suggests that drug changes during the grow out period of broilers may also influence the microbiome, and this change could be detrimental to the established microbiome. De Maesschalck (2015) ob- served that when a xylo-oligosaccharide (XOS) was fed to broilers, it stimulated the development of butyrate-producing bacteria, which are reported to have a beneficial effect on the animal. Other studies demonstrated an increase in Bifidobacterium in the intestine of broilers supplemented with XOS (Ribeiro et al., 2018).
Choct (2006), however, observed that supplementation of a xylanase enzyme reduced the digesta viscosity generated by feeding a high-soluble wheat and by reducing the viscosity a reduction in the content of total bacteria; specifically, Clostridium perfringens within the di- gesta also were observed.
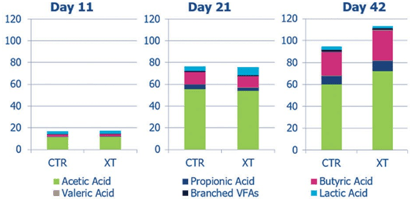
The production of VFAs is influenced by animal age, dietary fiber and enzyme usage. As the bird ages, the presence of an NSP enzyme in the diet leads to greater production of VFAs, particularly butyric acid (Figure 2). This is most like- ly due to the accelerated generation of xylo-oligosaccharides in the presence of the enzyme. VFA production at any age can reduce pH in the ceca and colon and serve as an energy source, stimulating enterocyte replication and mineral absorption and reducing osmotic diarrhea (Kumar et al., 2012).
Fermentation can also stimulate gut hormone production, such as enteroglu- cagon and PYY (Goodland et al., 1987), thereby reducing intestinal motility and feed intake in mammals (Chaudhri et al., 2006), allowing more time for digestion and absorption.
Several authors have seen an increase in the performance of broilers (Courtin et al., 2008; Suo et al., 2015) and swine (Liu et al., 2018) fed diets supplemented with XOS.
Bedford and Walk (2017) concluded that soluble, fermentable fiber, especially if viscous, can create problems for younger animals due to the disruption to digestion, resulting in a supply of excess carbohydrates and nitrogen to the large intestine, fueling dysbacteriosis and diarrhea. However, as the authors described, if viscosity is reduced through the use of an NSP enzyme, the same fiber can be beneficial to microbiome stability, with short-chain fatty acid production contributing to improved intestinal health.
Understanding fiber and its impact on animal physiology will be an important tool for nutritionists who are looking to improve gut function in the antibiotic- free era. The development of products that focus on stimulating lower gut fiber fermentation and enhancing beneficial microbiome growth will be of increased value for animal production.
These products may have different forms and act in multiple ways: specific bacteria that ferment fiber (probiotics), substrates to stimulate fiber fermentation in the lower gut (prebiotics), reduced viscosity effects and improved fiber solubility (xylanases) and providing products of fiber fermentation (pro- tected acids such as lactic and butyric).
A challenge to the nutritionist is under- standing how to utilize these products solely or in combination in order to develop a highly active fiber-fermenting microbiome early in the animal’s life from which the animal will benefit.
Reference available on request.