Tanmay Mondal1 and Gurpreet Singh2
Over 10 thousand years have passed since humans started farming different animals. Back then, people selected animals with certain traits to breed, a process that took many generations. Nowadays, farming is evolving quickly, and new challenges arise, like ensuring the safety of breeding methods and reducing harm to the environment.Traditionally, farmers would choose animals based on specific characteristics over generations. This process was time-consuming. In contrast, recent technologies like genome editing allow scientists to create animals with specific traits in just one step, using our knowledge of genes. This field, known as creating transgenic animals, is growing rapidly and offers many benefits in a short time.However, genome editing raises safety concerns compared to traditional breeding. Despite this, genetically modified farm animals, including cattle, sheep, goats, pigs, rabbits, chickens, and fish, have been successfully produced. Research in gene engineering of farm animals focuses on improving food production efficiency, animal health, and welfare. It also aims to reduce the impact of livestock on human health and the environment.
Gene-Edited Farm Animals in Medical Research
In medical research, gene editing of farm animals can be used for various applications, from producing proteins on a large scale to creating organs for transplantation. Some large animals serve as models for testing drugs, implants, or surgical procedures. The main strategy for genetically modified farm animals is to maintain breeding stocks, ensuring healthy herds with desired traits. These animals are then used for conventional breeding, and their offspring are used in production.Pioneering experiments include creating transgenic farm animals like knock-out pig and ferret models for CFTR, a gene related to Cystic Fibrosis. These larger animal models mimic human diseases more closely than smaller animals like mice, making them valuable in medical research.
Industrial Use of Transgenic Livestock
Boosting weight gain in beef cattle and pigs stands as a primary goal for the industry. An illustration of the enhanced efficiency of transgenic animals is seen in genetically modified (GM) gilts, which produce a remarkable 70% more milk than their non-transgenic counterparts. The offspring of these gilts experience a significant growth spurt, growing 500 grams larger in just 21 days of lactation.An alternative method to enhance productivity involves manipulating the reproductive performance of livestock. In one study, a point mutation was introduced into the GDF9 gene in goats, impacting ovulation rate and litter size.
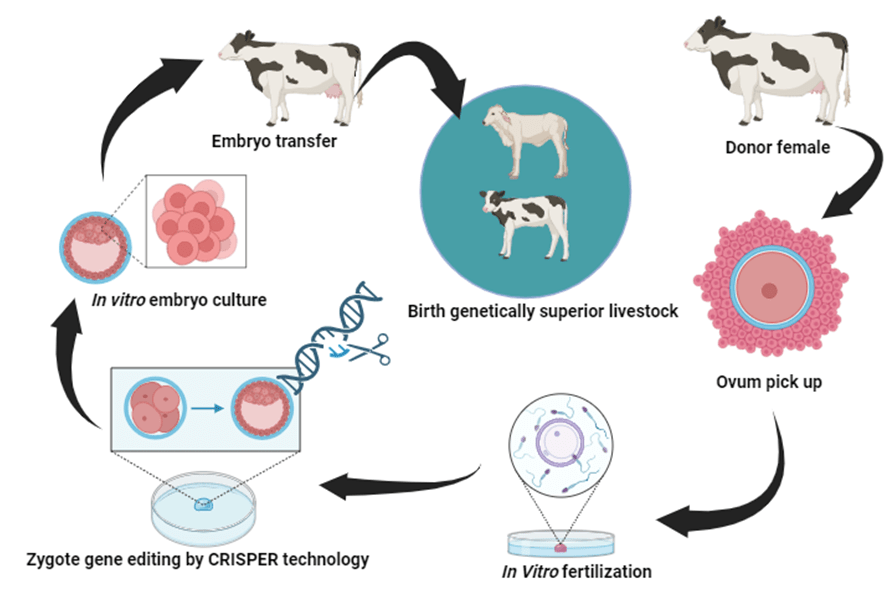
Figure 1.Methods of gene delivery applied in livestock.
Similarly, a mutation in the BMPR-1B (FecB) gene in sheep increased ovulation rates and subsequently enlarged litter sizes. Recently, researchers introduced a mouse gene regulating body temperature into pigs, enabling them to maintain an optimal temperature in cold weather by burning fat. This resulted in pigs with 24% less adipose tissue, offering the potential for low-fat pork production.Genome engineering addresses various needs in livestock maintenance. Transgenic goats with knock-out mutations in genes inhibiting hair and muscle growth enable increased production of cashmere and meat. Targeted mutagenesis using the CRISPR method has been applied to chicken genes encoding common egg allergens, with hopes of producing hypoallergenic eggs. Sheep and pigs are actively used for genome editing to mimic key features of human diseases, and large animals with specific mutations serve as models in physiology research.
Conclusions:Despite these promising advancements, the widespread adoption of transgenic animals in agriculture faces obstacles. Concerns about the biosafety of products from genetically modified (GM) animals and the high costs associated with obtaining and certifying transgenes hinder the integration of livestock biotechnologies into mainstream agricultural practices. While the potential is evident, the application of these technologies on a large scale remains a challenge in the near future.