Dr. Sumeet Singh and Dr. Manmeet Malpotra
Also known as Swine influenza / Pig influenza / Hog flu / Pig flu. The first reports of influenza like disease in pigs occurred in both US and Europe in conjunction with 1918 Human influenza pandemic. Human virus and H1N1 swine viruses were found to be related. Influenza viruses are one of the major causes of acute respiratory disease outbreaks in pigs, but infections are mostly subclinical. Epidemiology involves complex interplay of viruses of human, avian and swine origin.
Etiology and Morphological characteristics:
1) Members of family Orthomyxoviridae
2) Enveloped virus, which is susceptible to detergents, codes for 10-11 viral proteins on 8 segments of negative sense SSRNA.
3) Segmented nature of genome allows two viruses that co-infect a single host to exchange RNA segments during viral replication known as genetic reassortment.
4) Types of proteins found- Matrix protein (M), Nucleoprotein (Mainly A type found in swine viruses)
Reassortment between avian and mammalian viruses occur to produce a novel pandemic strain because swine tissue has both avian and human like sialic acid receptors. So, it is called as a “mixing vessel”. Swine viruses are also capable of infecting waterfowl, turkey etc.
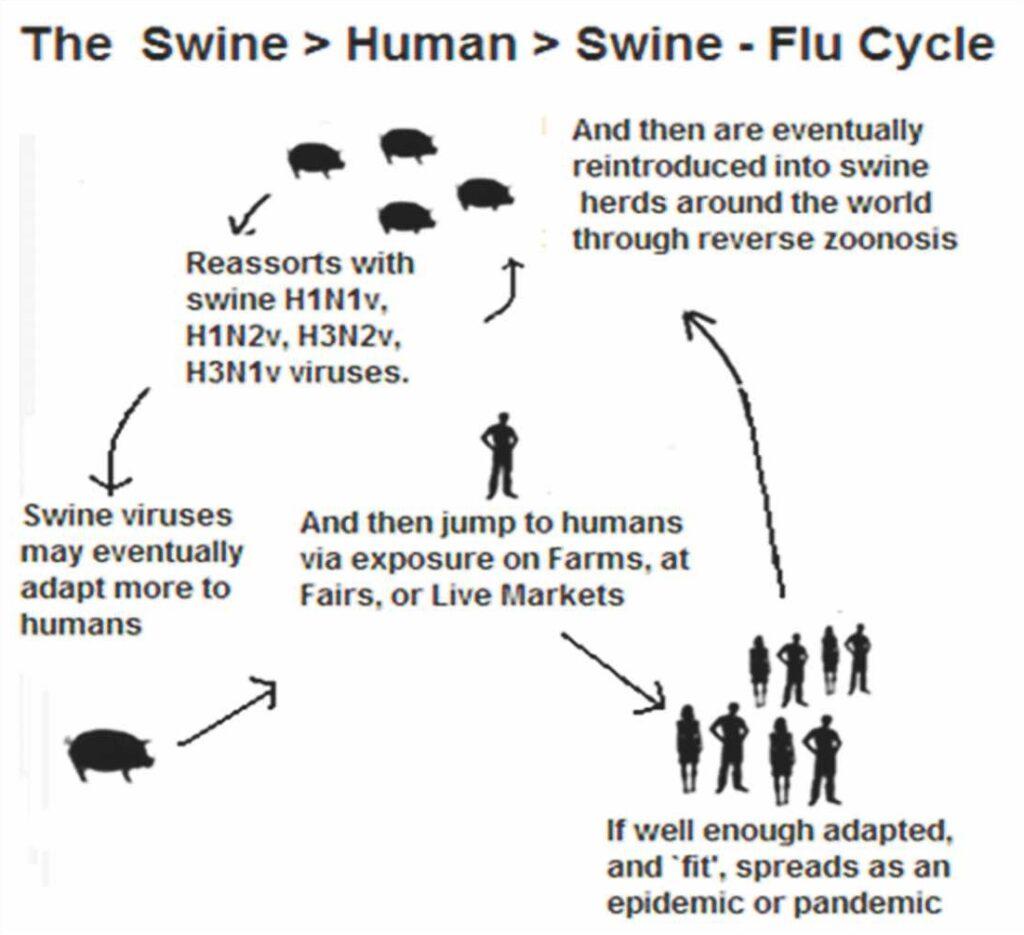
Koen in 1919 first noticed and observed similarities in disease with porcine and human. Martin Kaplan at WHO promoted the role of animals in epidemiology of influenza in humans at time of Asian Influenza pandemic in 1957. Dr Bernard Easterday firstly conclusively demonstrated transmission of swine influenza to farm personnel in 1976.
Transmission:
In northern climates, it occurs most frequently during the late fall through early winter months, often in association with the onset of colder temperatures and cold autumn rains. Influenza commonly introduced into herds through movement of animals. Primary route of virus transmission is through contact via nasopharyngeal exposure. Aerogenic transmission is there in highly populated areas with pig herds. Human to human transmission also occurs by sneezing.
Pathogenesis:
The virus multiplies in bronchial epithelium within 16 hr. of infection and causes focal necrosis of the bronchial epithelium, focal atelectasis, and gross hyperemia of the lungs. Bronchial exudates and widespread atelectasis, grossly appearing as plum-colored lesions affecting individual lobules of apical and intermediate lobes, are seen after 24 hr. The lesions continue to develop until 72 hr. after infection, after which the virus becomes more difficult to demonstrate. There is presence of neutrophils in lungs. Virus is unlikely to spread beyond respiratory tract. The brain stem is single extra respiratory tissue from which low amounts of virus is occasionally isolated.
Clinical Signs:
- Incubation period is 1-3 days
- Morbidity-100%
- Mortality-<1% (animals), 3-5% (human)
- Recovery- 5-7 days after onset
- A classic acute outbreak is characterized by high fever (to 108°F [42°C]), tachypnea, inactivity, huddling, coughing, labored abdominal breathing in animals. Decreased reproductive performance, increased infertility, abortion, stillbirths, small weak litter are also found.
- Pneumonia is the most common serious complication
- In humans’ signs observed are dyspnoea, weight loss, chills, dizziness, loss of appetite, fatigue.
Lesions:
Lesions usually are confined to the thoracic cavity
- Apical and cardiac lobes of lungs affected
- Interlobular edema, airway filled with blood-tinged fibrinous exudates, mediastinal lymph nodes enlarged
- Necrosis of lungs, presence of inflammatory cells in lungs
- Histologically, the lesions, when fully developed, are primarily those of an exudative bronchiolitis with some interstitial pneumonia.
Immune Response:
Rapid response to infection and results in complete elimination of virus from respiratory tract within 1 week. Antibodies detected 7-10 days post inoculation, peak at 2-3 weeks and decline by 8-10 weeks. Immune response with I/M administered killed vaccine is different from that with infection. Maternal derived antibodies can protect but decline after 4-14 weeks period.
Diagnosis:
A presumptive diagnosis can be made on clinical and pathologic findings, but confirmation depends on detection of viral RNA via RT PCR (Nasopharyngeal + Throat swabs), molecular sequencing, or demonstration of virus-specific antibody. Virus can be isolated from nasal secretions in the febrile phase or from affected lung tissue in the early acute stage .HI test can be used.
Prevention and control: There are no effective treatment, although antimicrobials may reduce secondary bacterial infections.
- Drugs like Oseltamivir @ 75 mg/kg b.i.d can be given for 5 days in humans
- Drugs like zanamivir, amatadine and rimantadine can also be used
Guidance for the States/ UTs on Seasonal Influenza Vaccination
- The recommendations for prioritized groups are as under: Health Care workers, working in hospital / institutional settings (doctors, nurses, paramedics) with likelihood of exposure to Influenza virus should be vaccinated. Vaccine is recommended for:
- Persons with chronic illnesses such as Chronic Obstructive Pulmonary Disease, Bronchial Asthma, Heart disease, Liver disease, Kidney disease, Blood disorders, Diabetes, Cancer and for those who are immunocompromised.
- For children having chronic diseases like Asthma; Neuro developmental condition like cerebral palsy, epilepsy stroke, mentally challenged etc.; heart disease like CHI), CHF; blood disorders like Sickle cell disease; diabetes, metabolic disorder, all immunocompromised children, malignancy receiving immuno-suppressive therapy, kidney disorder and liver disorder. Vaccine is desirable for
- Elderly individuals (≥ 65 years of age)
- Children between 6 months to 8 years of age.
- Two injections of vaccine 2-4 weeks apart with biannual booster vaccinations recommended for sows. Monovalent vaccine was first introduced in U.S. in 1993 of H1N1. After emergence of flue in 1998 by H3N2, multivalent vaccine was introduced. In humans, 2 doses of killed vaccine given 3-4 weeks apart, 0.5 ml (for age > 3 years), subcutaneous route
Outbreaks and Global Situation:
Spanish Flu (1918-1920) – It was the first outbreak, H1N1 was involved. It affected 500 million people and resulted in death of 20-50 million people. It was first seen in sUSA (Kansas area) and then it spread to France, Germany and UK. Newspapers were first to report in neutral Spain, gave false impression of Spain being severely hit and also King Alfonso XIII got infected of Spain ,so the name Spanish flu was used. Malnourishment, overcrowded medical camps, hospital, poor hygiene due to recent war promoted bacterial superinfection. In India alone 12-17 million people (5% of the total population) died due to this disease.
There were large number of deaths in different countries:
- Finland-20,000
- Sweden- 34,000
- Japan- 3,90,000
- Indonesia- 15,00,000
- New Zealand- 9,000
- Brazil- 3,00,000
- USA- 5,00,000 – 8,50,000
- England- 2,50,000
- France- 4,00,000
- Ghana- 1,00,000
- Russia- 4,50,000
- China 50,00,000-90,00,000
1968 Hong Kong Flu pandemic- H3N2 was involved in this pandemic. It evolved through reassortment of H1N1 virus. Otherwise upto 1997 only H1N1 was involved in diseases throughout the world.
2009 influenza pandemic- H1N1 was involved in this pandemic. Despite the fact that 2009 H1N1 virus was termed as swine flu, there is no evidence to suggest that infection in pigs epidemiologically contributed to widespread human transmission. It affected 2,84,000 people worldwide and caused death of 18500 humans. In India, 37000 cases were there out of which 1833 people died.
2015 & 17 outbreak in India: As of march 2015, the disease infected 31,151 people and took life of 1841 humans. Large deaths occurred in Delhi, MP, Rajasthan and Gujarat.
In 2017 there was outbreak in Maharashtra and Gujarat and 1090 people lost their lives in it.
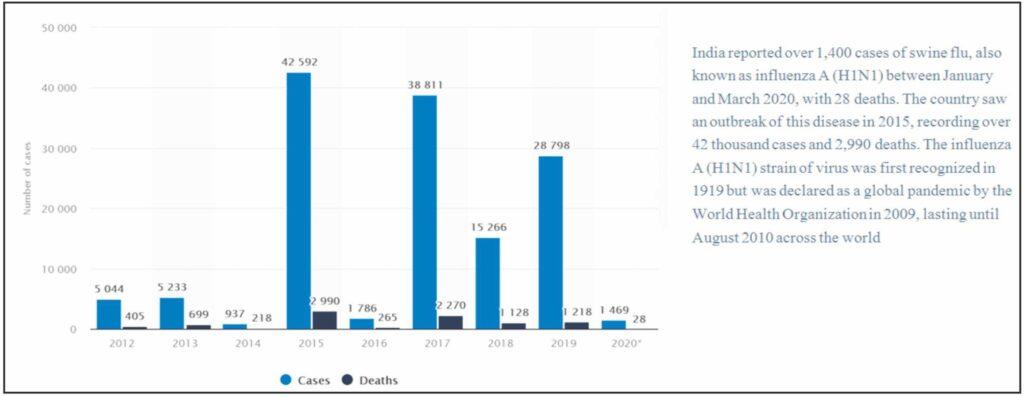
Other Outbreaks: Apart from this many small outbreaks were seen in different parts of the world with less causalities like in 2007 in Philippines, 2009 in Ireland, 2015 in Nepal, 2016 in Pakistan.
Economic losses:
It has caused a loss of 0.8% GDP of entire world.
India’s GDP also declined by 12.8% during Spanish flu pandemic
An estimated loss of $360 billion has been caused worldwide by this disease
References are available on request.